Metal Properties Database
(Pure and Alloyed)
A single crystal of any pure metal will have unique, very specific and consistent physical, electrical, chemical and mechanical properties that will always be identical no matter how, where, or when the tests are carried out and the measurements taken.
Work Hardening
On the other hand you can rarely create a single crystal of a 100% pure metal. It is extremely difficult to keep everything out and it will always have been handled prior to testing, which will almost certainly have disrupted a perfect crystal structure. Furthermore, even if you manage to create a single crystal of your metal, as soon as you try to perform mechanical tests on it, the single crystal will break up into many smaller crystals making it stronger. This is the basis for work hardening. As a metal's structure deforms, most of its properties will change. As a rule, the more crystals a given volume of material contains (i.e. the smaller they are) the stronger and more brittle the material will be.
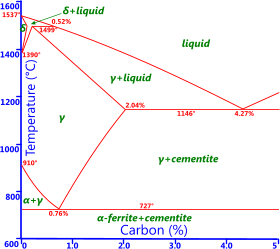
Fig 1. Iron-Carbon Phase Diagram
The problem with work hardening a pure metal to generate improved strength is that the material will gradually revert to larger crystals (reducing strength) over time with relatively modest temperature increases. The easiest way to ensure that small crystals remain small (retaining the metals' strength) is to add foreign elements that will lodge (or trap) themselves in the crystal (or grain) boundaries
Many properties of most pure metals (iron in particular) are quoted with a range of values due to the disparity of conditions at the time of testing.
Heat Treatment
All metals possess a natural state that is specific to a particular temperature range. For example, iron crystals sit naturally in a body centre cubic lattice form at room temperature, which is its 'alpha' state. Every metal goes through changes of state (or phase; e.g. alpha, beta, gamma, delta) as they are heated or cooled, above or below, very specific temperature boundaries indicated by the red lines in the example phase diagrams provided Fig 1 & Fig 2. If kept at a specific temperature, a metal will remain in that state forever. The point at which iron, for example, changes will depend upon the amount of carbon it contains. If you cool the metal below a temperature boundary rapidly enough you will lock-in an unnatural state, making it resistant to deformation (i.e. increasing hardness and strength).
You can of course reverse this condition. If you have a steel that is too hard and you want to soften it a little, simply heat the material to just below the upper temperature boundary for its alpha phase (for example), hold it at that temperature for a while depending upon the amount of softening you require, and let it cool down, again at a rate sufficient to allow just the right amount of re-crystallisation to occur.
The problem with the above technique is that if during the metal's working life it is periodically exposed to high temperatures...
a) ... below the upper boundary it will continue to soften over time
or
b) ... above the upper boundary it will harden over time
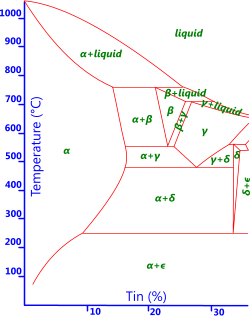
Fig 2. Copper-Tin Phase Diagram
Alloying Elements
An alloying element is the intentional addition of another (different) pure element, evenly distributed and in sufficient quantity to suitably alter the base metal's properties. Whilst work hardening (see above) and heat treatment (see above) can alter the base material's strength, hardness and toughness (ductility), they cannot modify its chemical properties; temperature and corrosion resistance, magnetism, colour, etc. Alloying elements can do both, and the properties created endure for the life of the alloy.
The type/level of benefit(s) will vary with the structure of the alloy, which can be controlled/altered by varying the quantity of the alloying element. An obvious example of this technique is stainless steel where the addition of various significant amounts of elements such as chromium and/or nickel will alter the basic ferrite structure of iron to austenite, pearlite or martensite or a combination (Duplex) significantly improving corrosion resistance, whereas lesser amounts would simply have altered strength, hardness and ductility.
Iron & Steel
Despite the continuing discovery of new and improved materials, steel remains the most versatile and cost effective of them all. You (or your forging company) can take almost any steel and turn it into something you need by working it, heat treating it or adding alloying elements. So you needn't worry if you can't find exactly the properties you're looking for, just select the closest specification and ask your forging company to modify it for you.
You can usually comply with a recognised or accepted steel standard or specification because alloying elements are always specified as maximums or ranges and you can considerably modify/improve its mechanical properties by making small changes to a combination of its chemical composition, heat treatment and cold working.
In addition to its structural, chemical and temperature properties, when designing an alloy you should also consider the likelihood of welding where a high 'Carbon Equivalent' (CEV) will result in localised embrittlement. CEV is calculated using either of the following two formulas:
Low & medium carbon steel: CEV = C + Mn/6 + (Cr+Mo+V)/5 + (Ni+Cu)/15
or
High carbon steel: CEV = C + (Mn+Si)/6 + (Cr+Mo+V)/5 + (Ni+Cu)/15
Where you enter the actual percentage of the relevant element your steel contains in the appropriate part of the formula. The aim is to keep CEV below 0.45 for reasons of toughness and below 0.39 for reliable welding.
One of the most commonly available steels used for the forging of structural beams (H-Beams, I-Beams, Channel, Angle, etc.) is S275JR (formerly BS 4360-43B), the properties of which are included in CalQlata's Metals database.
See Carbon Steels for a more detailed description of carbon steels.
Steel Alloys
Each element affects one or more physical property of any base metal in a particular way. For example:
Carbon will get between the grains of iron preventing crystal growth, thereby maintaining the material's properties throughout its life. Steel is iron mixed with carbon. The more carbon it comprises, the smaller the iron grains, the harder the steel and higher its strength.
Nickel will add hardness and strength but without significantly affecting ductility.
Chromium will add hardness and strength and significantly reduce ductility. However, it improves wear resistance and refines grain structure.
Manganese improves strength and toughness greater than Nickel and Chromium.
Vanadium increases strength and toughness and greatly increases and improves (better penetration throughout the metal) hardness.
Molybdenum has a similar effect to Nickel but with even lower loss of ductility. These steels generally have highest strength and toughness with greatest elongation and they can even be machined when hard.
Silicon increases resistivity and greatly reduces hysteresis loss so it is very useful for electrical transformer cores.
Very small amounts of Boron (<0.001%) will significantly improve (better penetration throughout the metal) hardenability of a high-carbon steel. You can recognise a Boron steel because it has a letter 'B' in the middle of its AISI specification (e.g. 8145 becomes 81B45).
Stainless Steel
The principal problem for carbon steel is degradation of its properties over time from corrosion or any other chemical attack. For this reason, alloying elements are added in sufficient quantity to minimise or even prevent it – i.e. the creation of Stainless Steel. The problem is that corrosion protection must differ according to the form of chemical attack expected. All stainless steels have iron as their base metal and are generally grouped according to its phase structure within the alloy:
Ferritic: Contains chromium ≈18% which is increased to ≤30% for high temperature corrosion resistance. Hardened by cold working, not through heat treatment. Always magnetic. Relatively low cost corrosion resistant stainless steel. Low carbon content hence extremely ductile. Can be heat treated and/or cold worked. Used for kitchen and dairy equipment, decoration and car trimming.
Austenitic (18-8): Contain ≈18% nickel & ≈8% chromium. Does not heat treat (except for annealing) but hardens through cold working. Non-magnetic when annealed. Good machinability, can be welded and easily forged into complex shapes. Excellent high temperature corrosion and acid resistance. Good low temperature toughness. Susceptible to sulphide stress corrosion cracking.
Martensitic: Contains chromium ≈12% and carbon content can be increased more than in the other two. Can be heat treated to high hardness. Usually magnetic. Normally used for razor blades, knives and instruments because it keeps sharp edge. Particularly resistant to fruit and vegetable acids.
Duplex: A mixture of Austenitic and Ferritic stainless steel in equal proportions. Excellent high temperature strength and resistance to hydrogen sulphide stress corrosion cracking and pitting resistance. Normally used for components exposed to highly corrosive fluids and gasses at high temperature, e.g. the petroleum industry.
Copper & its Alloys
Unlike all other alloying elements, the quantities of the special elements of tin (Sn) and zinc (Zn) in these alloys are actual values and not maximums. It is the actual values of these alloying elements that define the properties of each copper alloy.
Copper alloys and brasses can be work hardened or heat treated to provide excellent strength, corrosion resistance along with excellent machining and forming properties.
More expensive than most steels (including stainless and alloys) due to the cost of copper.
Cannot reach the strength or hardness of the better steels.
These are grouped as follows (Cu+):
Copper Alloy (+Zn); Good conductor of electricity and heat and resists oxidisation. High-alloy components also have high hardness and can be used for gears, bushes, bearings, etc.
Brass (+Zn); Plumbing, general purpose and ornamental goods.
Phosphor Bronze (+Sn); Excellent resistance to steam. Piston rings, springs, pump impellers, bushes and bearings.
Aluminium bronze (+Al); Good seawater resistance with high strength and machinability. Bearings, bushes, gears, etc.
Copper-Nickel (+Ni); Best resistance to seawater of all brasses/bronzes
Silicon Bronze (+Si); Bells, propellers and intricate die castings.
Manganese and Leaded Bronze (+Pb); High-strength components for engine parts and marine equipment (e.g. high-strength racing propellers) and wear guides.
Nickel-Silvers (+Ni & Ag); Musical instrument, ornamental and hardware fittings.
See Copper Alloys for a more detailed description of copper alloys.
Titanium & its Alloys
Titanium is a light-weight alternative to steel and high-strength alternative to aluminium and more expensive than both. In fact it has a better strength to density ratio than any other metal. However, titanium and its alloys have a tensile modulus of about half that of steel, which means that for the same load, it will elongate twice as much. This may or may not be acceptable for many applications, but for everyday use such as in bicycle frames it is not working hard enough for stress-growth to occur.
In alloyed form, titanium has the same strength as high-strength steels but almost half its density, so you can manufacture something of equal strength but half the weight if you can live with additional elongation. Everything is a compromise.
Aluminium & its Alloys
Some specialist (and expensive) aluminium alloys will give you the strength of a mid-range steel (500 MPa) for just a third of the density. But it also has a tensile modulus a third of that of steel so you get three times the elongation for the same stress.
Aluminium quickly self generates a thin oxide skin which provides it with excellent corrosion resistance. If this skin is removed in part or in full, however, it will corrode quite quickly.
Aluminium is a good thermal and electrical conductor but is resistant to magnetism and is sometimes used as a magnetic shield.
These alloys lose some of their strength more quickly than steel and titanium at elevated temperatures but retain their ductility at very low temperatures, though less well than the best (and more expensive) stainless steels.
Nickel & its Alloys
Nickel is similar to iron in many respects but with much better corrosion resistance. It also accepts and responds well to many alloying elements, so you can do almost anything with this metal. The resultant alloys can show exceptional resistance to oxidisation and chemical attack and provide strength almost as good as the best alloy steels. You can machine, form, weld or cast nickel. Nickel alloys are generally grouped as follows:
Nickel alloys (Ni>94%): Ductile and tough to cryogenic temperatures with good electro-magnetic properties
Inconel: nickel-chromium-molybdenum for strength and corrosion resistance in extreme conditions
Incolloy: nickel-chromium-iron for high strength and corrosion resistance in elevated temperature conditions
Hastelloy: nickel-molybdenum for machinability
Monel: nickel-copper for actively corrosive environments and higher ductility than Inconel
Magnesium & its Alloys
Magnesium has a quarter of the density of steel and up to a quarter of its strength, but it elongates five times as much as steel for the same stress. So whilst you cannot achieve any greater strength to weight ratio than steel, used correctly in components that require bulk (e.g. vehicle wheels) a significant weight saving is possible. There are limits to its prowess in wheels, however, growth under stress is probably the reason why it would not be wise to use magnesium wheels on Noble's land speed vehicle Bloodhound SSC where they have used a very high-strength aluminium alloy.
Magnesium dust can also be hazardous in a working environment and is combustible at high temperatures, which can be readily generated during machining.
Precious Metals
Palladium, platinum, gold, silver, etc. are no longer simply used only for decorative jewellery, they are also essential for many every-day applications such as televisions, computers, mobile telephones and car parts. The problem is these are very rare metals (hence the reason why they're precious) and they work best in their pure form, so you need more of it. They are rarely suitable for manufacturing components (other than jewellery) and are usually plated onto other materials, so their electrical properties are important.
Metal Properties Database - Technical Help
Applicability
Metals' database is applicable specifically to those metals included within it, but generally applicable to all metals and their alloys. For example, there are a number of steels and iron alloys included in the database, the properties of which apply specifically to the associated AISI grade. But if you require a material with different properties to those available within the grades you see in the Metals database, you simply pick the nearest grade and ask your local forging company to modify it to meet your specific requirements. He/she will normally be able to do it fairly easily. You simply make sure that the material you are eventually provided with complies; a) with the specification and; b) retains an acceptable carbon equivalent if it needs to be welded.
You can harden any carbon steel on its surface or right through if it has the correct alloying elements. You can tell how suitable your metal is for hardening by its 4-digit AISI number:
1016 to 1030 low carbon: are materials of high ductility that can be case hardened (outside hard and inside tough)
1031 to 1052 medium carbon: higher strength than above and case harden to a greater depth
1055 to 1095 high carbon: much higher strength, less ductility and hard to a much greater depth
If the second digit in the AISI number of a carbon steel is a '1' (e.g. 1131) it means the material is the same as the material with '0' (e.g. 1031) but with alloying elements like lead or copper to make it more machinable
High-strength/low-alloy steels begin with a '9' and contain only 3 digits (e.g. SAE 950).
Apart from '1330' to '1345' (manganese steels), the first digit in the AISI designation for alloy steels is '4' or above.
Through-hardening steels, i.e. those that can heat treated to provide that same hardness through out the body of the material, are alloy steels.
Application
In almost all cases, elongation decreases with increasing strength and vice-versa.
Many carbon steels have a range of mechanical properties that will vary according to their thickness. The thicker the material is, the lower its strength but also the greater its elongation and its toughness.
The chemical composition of any alloy metal as specified in Metals is as follows:
Single value; C:0.47 means that the element concerned (e.g. carbon) can be anything from zero to the percentage quoted
Value range; C:0.23<0.39 means that the element concerned (e.g. carbon) can be anything from the smallest percentage quoted (e.g. 0.23%) to the largest percentage quoted (e.g. 0.39%).
Validity
The values quoted in Metals are believed correct by CalQlata from its own historical information. There are many sources of information for the grades quoted above and most vary to a greater or lesser extent. As such, CalQlata would recommend that you perform those calculations you need using the material specifications in the database and then go directly to the specification and/or your local forging company to finalise all your material requirements and ensure specification compliance.
Further Reading
You will find further reading on this subject in reference publications(1, 2, 3, 12 & 17)

